The debate around tirzepatide vs semaglutide has become one of the most searched health topics in the US, with over 44,000 monthly searches and growing. Both medications have transformed weight loss and diabetes treatment, but they aren't the same drug, and they don't work the same way.
Semaglutide (sold as Ozempic, Wegovy, and Rybelsus) arrived first and built the largest body of clinical evidence for any GLP-1 medication. Tirzepatide (Mounjaro for type 2 diabetes, Zepbound for obesity) followed with a dual-hormone mechanism that has consistently outperformed semaglutide in head-to-head trials.
So which one actually delivers better results? And more importantly, which one makes sense for a specific patient's body, budget, and health goals? Both rank among the most effective peptides for weight loss available today.
This guide breaks down the clinical data, side effect profiles, dosing schedules, costs, and practical decision factors, all based on published trial results and real-world outcomes through early 2026. No hype. Just what the evidence says.
How Tirzepatide and Semaglutide Work Differently in the Body
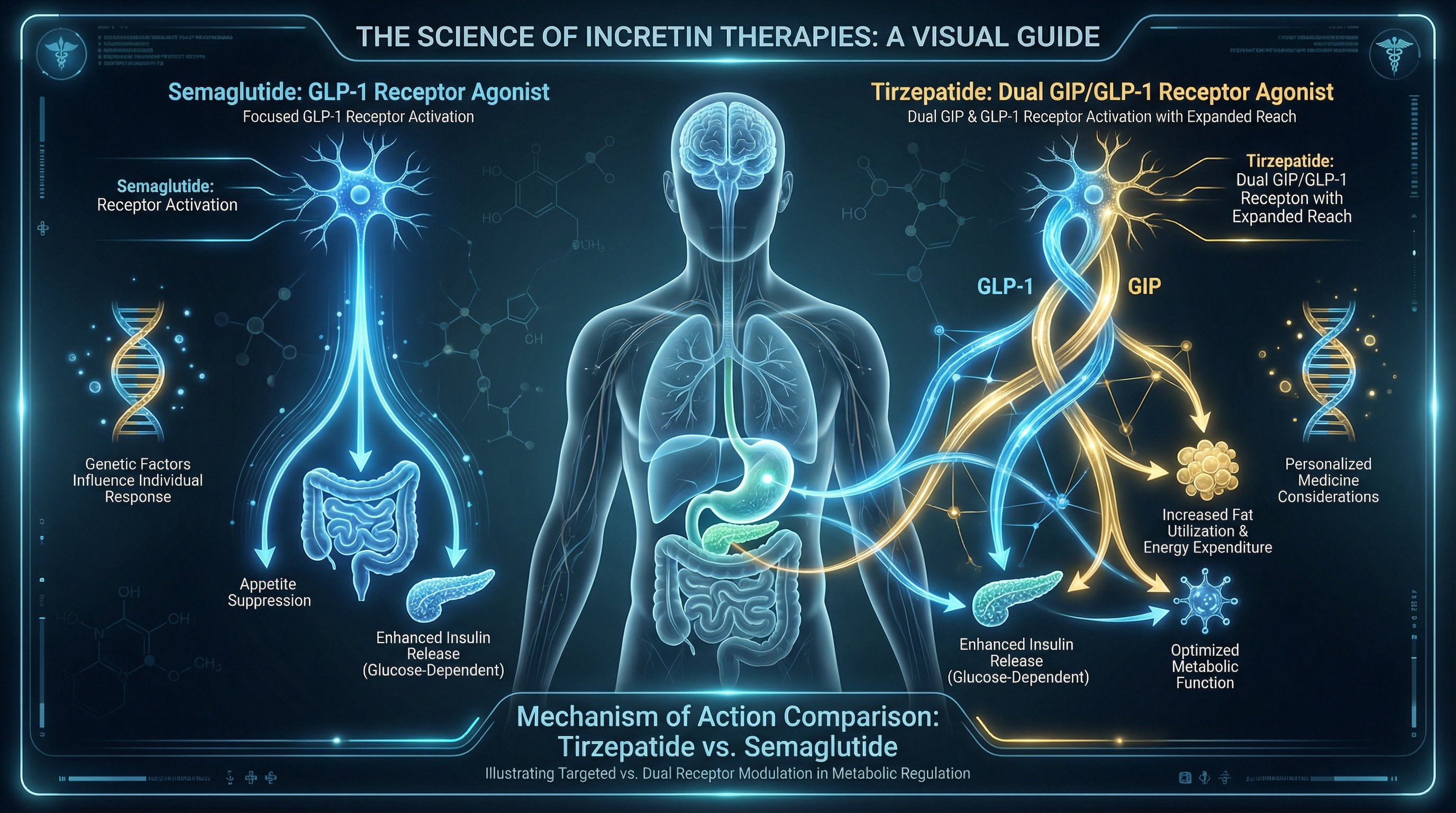
Understanding the mechanism behind each drug explains why their results differ.
Semaglutide is a GLP-1 receptor agonist. It mimics a single gut hormone, glucagon-like peptide-1, that signals the brain to reduce hunger, slows gastric emptying, and stimulates insulin release when blood sugar rises. This single-target approach made semaglutide the gold standard for GLP-1 therapy for several years.
Tirzepatide is a dual GIP and GLP-1 receptor agonist, the first of its kind approved by the FDA. It activates two incretin hormone pathways simultaneously:
- GLP-1 receptor: Reduces appetite, improves insulin secretion, slows stomach emptying
- GIP receptor: Enhances fat metabolism, improves insulin sensitivity, and may contribute additional appetite suppression through pathways GLP-1 alone doesn't reach
This dual mechanism is the core reason tirzepatide consistently produces greater weight loss and HbA1c reductions in clinical trials. The GIP pathway adds a metabolic dimension that pure GLP-1 agonists miss.
Genetics play a role here, too. Variants in the GLP1R gene (rs6923761) affect how strongly a patient responds to the GLP-1 component shared by both drugs. Patients with GLP1R A/A genotype tend to lose 3–5% more body weight. Meanwhile, GIPR gene variants (rs10423928) influence sensitivity to tirzepatide's second mechanism, which may explain why some patients respond dramatically better to tirzepatide than semaglutide.
Patients carrying the TCF7L2 T allele, associated with impaired GLP-1 signaling, may particularly benefit from tirzepatide's dual agonism since the GIP pathway can partially compensate for weaker GLP-1 receptor response.
Both drugs share the same basic delivery: subcutaneous injection, once weekly. But their biological fingerprints inside the body are meaningfully different.
Weight Loss and Blood Sugar Results: What the Clinical Data Shows
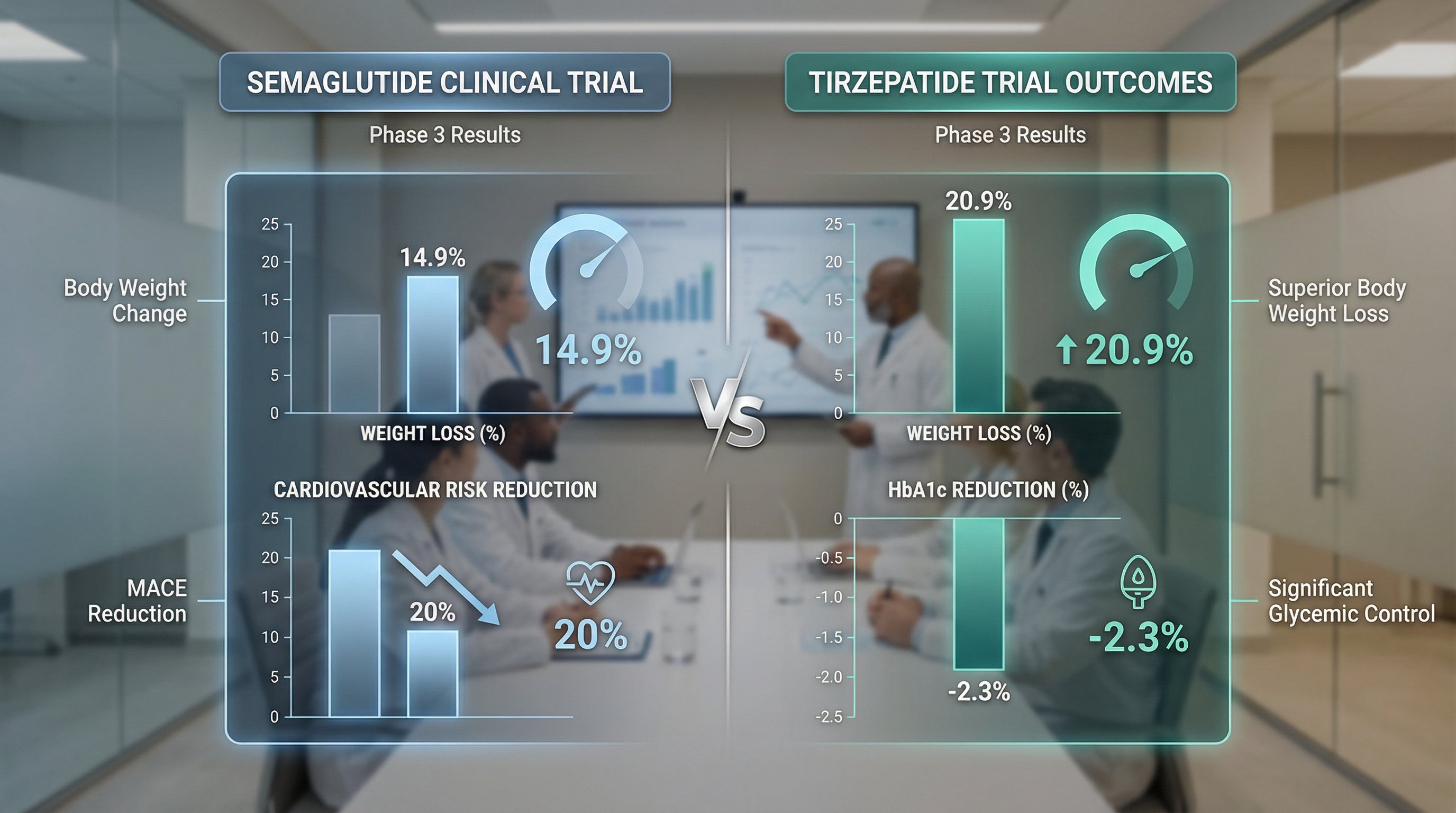
Numbers tell the real story here. Both medications have extensive Phase 3 trial data, and tirzepatide leads in nearly every efficacy measure.
Semaglutide Weight Loss Data
The STEP trial program established semaglutide's credentials:
- STEP 1: 14.9% mean weight loss vs 2.4% placebo at 68 weeks (1,961 patients)
- STEP 5: Weight loss maintained at 104 weeks with continued treatment
- SELECT trial: 20% reduction in major adverse cardiac events (HR 0.80, p<0.001) across 17,604 patients, the strongest cardiovascular outcomes data for any GLP-1 drug
Semaglutide's cardiovascular evidence is a major differentiator. The SELECT trial proved it doesn't just help people lose weight, it reduces heart attacks, strokes, and cardiovascular death.
Tirzepatide Weight Loss Data
The SURMOUNT trials raised the bar:
- SURMOUNT-1: 20.9% mean weight loss at the 15 mg dose vs 3.1% placebo over 72 weeks (2,539 patients)
- 36% of patients on 15 mg achieved ≥25% total body weight loss
- SURPASS-2: Superior to semaglutide 1 mg for both HbA1c reduction and weight loss in a direct head-to-head comparison
- HbA1c reductions ranged from 1.87% to 2.58% across doses in type 2 diabetes trials
Head-to-Head Comparison
A meta-analysis covering 142,811 patients from randomized controlled trials and real-world cohorts found tirzepatide produced a mean difference of 4.23% greater weight loss (95% CI: 3.22–5.25) compared to semaglutide. At doses above 10 mg, that gap widened to 6.50%.
Real-world data at 12 months paints a similar picture: -11.4% body weight with tirzepatide vs -6.2% with semaglutide.
Blood Sugar Control
Both drugs significantly reduce HbA1c in patients with type 2 diabetes. Semaglutide typically achieves reductions of 1.5–1.8%. Tirzepatide's dual mechanism pushes HbA1c down by 1.87–2.58%, making it the stronger option for patients with higher baseline HbA1c levels (above 8%).
For glycemic control specifically, SURPASS-2 showed tirzepatide was statistically superior to semaglutide at every dose tested.
Bottom line: Tirzepatide wins on raw efficacy. Semaglutide wins on cardiovascular outcome evidence. Both produce clinically meaningful results. Patients on either medication should pair therapy with a structured GLP-1 diet plan to maximize outcomes.
Side Effects and Safety Profiles Compared
The side effect profiles of tirzepatide and semaglutide overlap significantly. Both are incretin-based therapies, so they share the same core gastrointestinal side effects.
Common Side Effects
| Side Effect | Semaglutide | Tirzepatide |
|---|---|---|
| Nausea | 44% | Up to 33% |
| Diarrhea | 30% | 23% |
| Vomiting | 24% | Moderate |
| Constipation | 24% | Up to 25% |
| Decreased appetite | Common | Common |
One notable pattern: semaglutide tends to produce slightly higher nausea rates (44% vs ~33% for tirzepatide). This may surprise people who assume the dual-mechanism drug would cause more GI distress. The difference likely relates to tirzepatide's more gradual GLP-1 receptor activation.
Most GI side effects with both drugs are dose-dependent and temporary, they peak during dose escalation (see the semaglutide dose chart for titration details) and ease as the body adjusts. Escalating too quickly is the most common cause of severe nausea with either medication.
Serious Safety Concerns
Both medications carry the same boxed warnings and rare risks:
- Medullary thyroid carcinoma (MTC): Both carry a box warning based on animal studies. Patients with personal or family history of MTC or MEN2 syndrome should not use either drug.
- Pancreatitis: Rare but reported with both. Baseline amylase and lipase testing is recommended.
- Gallbladder events: Increased incidence reported across GLP-1 class medications.
- Hypoglycemia: Risk increases when combined with insulin or sulfonylureas.
Long-Term Safety Track Record
Here's where semaglutide has a clear advantage: more years of real-world use and larger safety datasets. The SELECT trial alone followed 17,604 patients and demonstrated not just safety but active cardiovascular benefit, including a 26% reduction in major cardiac events (SUSTAIN-6) and a 39% reduction in non-fatal stroke in type 2 diabetes patients.
Tirzepatide's cardiovascular outcomes trial is still pending. The drug is safe based on available data, but it hasn't yet matched semaglutide's depth of long-term evidence.
Monitoring recommendations for both drugs include:
- HbA1c + fasting glucose at baseline and 12 weeks
- Lipid panel at baseline and 12 weeks
- Thyroid panel (TSH, fT4) at baseline
- Amylase + lipase at baseline
- Comprehensive metabolic panel at baseline
Neither drug is appropriate during pregnancy or breastfeeding.
Dosing, Administration, and Cost Considerations

Dosing Schedules
Both medications require gradual dose escalation to minimize side effects.
Semaglutide (Wegovy/Ozempic):
- Starting dose: 0.25 mg/week
- Titration: Escalate every 4 weeks → 0.25 → 0.5 → 1.0 → 1.7 → 2.4 mg
- Maintenance: 2.4 mg/week (Wegovy) or 2.0 mg/week (Ozempic)
- Also available as daily oral tablet (Rybelsus), taken fasted with ≤4 oz water, 30 minutes before food
Tirzepatide (Zepbound/Mounjaro):
- Starting dose: 2.5 mg/week
- Titration: Escalate every 4 weeks → 2.5 → 5 → 7.5 → 10 → 12.5 → 15 mg
- Maintenance: 5–15 mg/week (efficacy is dose-dependent)
Both are administered via pre-filled pen, subcutaneously, once weekly on the same day each week at any time of day, with or without food. Missed doses should be taken within 4 days of the scheduled date: otherwise, skip to the next dose.
Storage: Refrigerate both medications. They can be kept at room temperature (≤30°C / 86°F) for up to 21 days if unopened.
The Oral Option
Semaglutide holds a unique advantage here: it's available as an oral tablet (Rybelsus). For patients who genuinely cannot tolerate injections, this matters. Tirzepatide has no oral formulation currently available.
Cost Realities
Neither drug is cheap without insurance. Brand-name pricing for both medications runs over $1,000/month at retail, though actual out-of-pocket costs vary widely based on insurance coverage, manufacturer savings programs, and pharmacy.
A few cost considerations worth noting:
- Semaglutide has been on the market longer, so insurance formulary coverage tends to be more established
- Tirzepatide often requires prior authorization, especially for the Zepbound (obesity) indication
- Generic liraglutide (Saxenda/Victoza) became available in 2025 and offers a budget-friendly GLP-1 alternative at roughly ~8% weight loss, far less effective, but a fraction of the cost
- Neither tirzepatide nor semaglutide has generic versions available yet
Patients should verify formulary status with their insurer before committing. The difference between a $25 copay and $1,300 out-of-pocket can change which drug makes practical sense.
For those exploring peptide therapy options and need help connecting with qualified providers, platforms like Peptide Injections use AI-powered matching to connect patients with board-certified physicians specializing in these treatments, often in under two minutes.
How to Decide Which Medication Is Right for You

Choosing between tirzepatide and semaglutide isn't about picking the "better" drug. It's about matching the right medication to a specific patient's profile.
Choose Semaglutide If:
- Cardiovascular risk reduction is a priority. The SELECT trial's 20% reduction in major cardiac events is unmatched. No other GLP-1 drug has this level of CV evidence.
- An oral option is preferred. Rybelsus eliminates injections entirely.
- Insurance coverage favors it. Semaglutide's longer market presence often means better formulary positioning.
- The patient is a strong GLP1R responder. Those with GLP1R A/A genotype may achieve excellent results with a single-mechanism drug.
- Age considerations matter. Semaglutide is FDA-approved for patients aged 12 and older for weight management.
Choose Tirzepatide If:
- Maximum weight loss is the primary goal. 20.9% mean loss (SURMOUNT-1) vs 14.9% (STEP 1), that gap is clinically significant.
- HbA1c is above 8%. Tirzepatide's dual mechanism produces stronger glycemic reductions across all doses tested.
- The patient is a partial GLP-1 responder. GIPR activation provides a second pathway. Patients with GLP1R low-response genotypes or TCF7L2 T carriers often do better on tirzepatide.
- Appetite-driven eating is the dominant pattern. The dual hormone mechanism may provide stronger satiety signals for some individuals.
- Previous GLP-1 therapy plateaued. Switching from semaglutide to tirzepatide has shown additional weight loss in real-world practice.
The Role of Genetic Testing
This is where the conversation is heading in 2026. Pharmacogenomic testing can identify variants in GLP1R, GIPR, MC4R, FTO, and TCF7L2 that directly influence which drug will produce the strongest response.
For example:
- MC4R risk carriers may plateau earlier on either drug
- FTO A allele carriers (higher obesity predisposition) tend to benefit more from strong appetite suppression
- TCF7L2 T carriers with impaired incretin biology may specifically need tirzepatide's dual mechanism
Genetic testing isn't required to start treatment, but it can prevent months of trial-and-error for patients who don't respond as expected to their first medication.
What About Newer Options?
Two emerging therapies are worth watching:
- CagriSema (cagrilintide + semaglutide): Achieved 20.4% weight loss in the REDEFINE 1 trial. Combines amylin and GLP-1 pathways in one injection. FDA decision expected in 2026.
- Retatrutide (triple GIP/GLP-1/glucagon agonist): 28.7% mean weight loss in TRIUMPH-4. The most powerful obesity drug tested to date. Earliest approval estimated mid-2027.
These aren't available yet outside clinical trials, but they signal where metabolic therapy is heading.
Talk to a Specialist
Both tirzepatide and semaglutide require a prescription and medical supervision. Patients should work with a physician who understands metabolic therapy, monitors bloodwork at appropriate intervals, and adjusts dosing based on individual response. Peptide Injections can match patients with specialized providers who focus on peptide therapy protocols, a useful starting point for anyone unsure where to begin.
Conclusion
Tirzepatide and semaglutide are both proven, FDA-approved medications that produce meaningful weight loss and blood sugar improvement. The data is clear: tirzepatide delivers greater weight loss (roughly 4–6% more than semaglutide), while semaglutide holds the strongest cardiovascular outcomes evidence available for any GLP-1 therapy.
Neither drug is universally "better." The right choice depends on individual health goals, genetic response profile, insurance coverage, and whether cardiovascular protection or maximum weight loss takes priority.
What's changed in 2026 is that patients don't have to guess. Between published trial data, real-world outcomes, and emerging pharmacogenomic testing, there's more information available than ever to make a confident, evidence-based decision with the right medical team.
Frequently Asked Questions About Tirzepatide vs Semaglutide
What is the main difference between tirzepatide and semaglutide?
Semaglutide is a single GLP-1 receptor agonist targeting one hormone pathway, while tirzepatide is a dual GIP/GLP-1 receptor agonist targeting two pathways simultaneously. This dual mechanism allows tirzepatide to activate both appetite suppression and fat metabolism pathways, contributing to greater weight loss results.
How much more weight can I lose with tirzepatide vs semaglutide?
Head-to-head clinical data shows tirzepatide produces approximately 4–6% greater weight loss than semaglutide. SURMOUNT-1 reported 20.9% mean weight loss with tirzepatide at 15mg versus 14.9% with semaglutide at highest doses, a clinically significant difference for severe obesity.
Does semaglutide have better cardiovascular evidence than tirzepatide?
Yes. The SELECT trial demonstrated semaglutide reduced major adverse cardiac events by 20% (HR 0.80) in 17,604 patients. Tirzepatide's cardiovascular outcomes trial is still pending, making semaglutide the only medication with proven long-term cardiac protection data available now.
Which medication causes more nausea, tirzepatide or semaglutide?
Semaglutide tends to cause higher nausea rates (44% vs ~33% for tirzepatide). Both medications share similar GI side effects including diarrhea and constipation, though most effects are dose-dependent and temporary, peak during escalation, and improve as the body adjusts.
Can genetics help determine if I should take tirzepatide or semaglutide?
Yes. Genetic variants in GLP1R, GIPR, and TCF7L2 directly influence medication response. Patients with GLP1R A/A genotype may lose more weight on semaglutide, while TCF7L2 T carriers with impaired GLP-1 signaling often respond better to tirzepatide's dual mechanism. Genetic testing at thepeptidelist.com can guide choice.
Is there an oral option available for tirzepatide or semaglutide?
Only semaglutide offers an oral tablet option called Rybelsus, taken daily while fasted. Tirzepatide is available only as a weekly subcutaneous injection. This oral alternative is significant for patients who cannot tolerate injections or prefer daily dosing control.